
EMBO J 10:25–33īennett MJ, Schlunegger MP, Eisenberg D (1995) 3D domain swapping: a mechanism for oligomer assembly. J Biol Chem 277: 4024–4033īeese LS, Steitz TA (1991) Structural basis for the 3’–5’ exonuclease activity of Escherichia coli DNA polymerase I: a two metal ion mechanism. Curr Opin Struct Biol 5:11–19īath AJ, Milsom SE, Gormley NA, Halford SE (2002) Many Type IIs restriction endonucleases interact with two recognition sites before cleaving DNA. This process is experimental and the keywords may be updated as the learning algorithm improves.Īggarwal AK (1995) Structure and function of restriction endonucleases. These keywords were added by machine and not by the authors. Hence, the symmetry of the recognition sequence implies the oligomeric state of the restriction enzyme. Symmetrical nicking of opposite DNA strands by both monomers within a homodimer generates the double-strand break. Each subunit faces the same nucleotide sequence on the opposite DNA strand and contains one active site. 1), recognition of the palindromic DNA sequence is achieved by two identical protein subunits related by a twofold axis of symmetry. On the basis of this observation, Kelly and Smith proposed the first model for the interaction of these restriction enzymes with DNA ( Kelly and Smith 1970).According to their model (Fig. e., possess a twofold rotational axis of symmetry. Most of the sequences recognized by Type II restriction endonucleases are palindromic, i. Around 3500 species, from variety of bacteria with nearly 240 differing specificities, have now been characterized ( Roberts et al. Moreover, even very few examples that we have examined so far indicate the importance of further studies on protein palindromes.Type II restriction endonucleases recognize specific DNA sequences, typically 4–8 bp in length, and cleave phosphodiester bonds in the presence of Mg 2+, within or close to their recognition sites ( Pingoud and Jeltsch 2001). 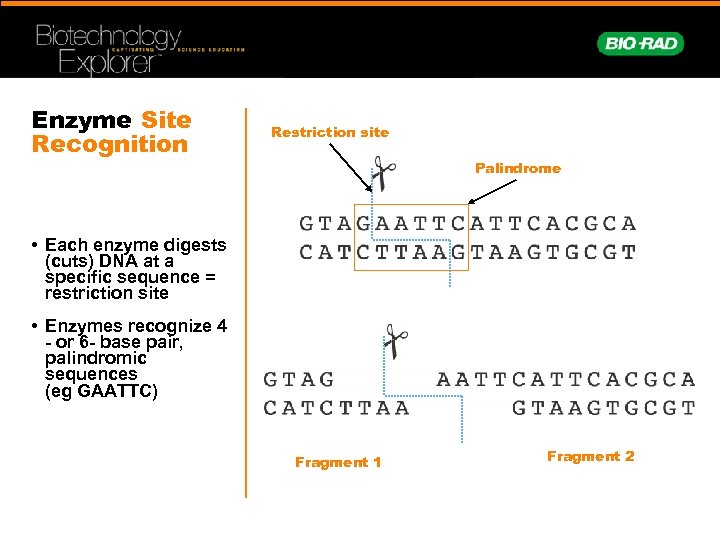
Our results have clearly showed that palindromes are frequently occurring motives in proteins. As the number of possible palindromic sequences of a given length is far much greater for proteins (20N) than for nucleic acids (4N), the study on their role seems to be an exciting challenge. Oligonucleotide LTI-ITL has been observed in the crystal structure and is located close to a DNA recognizing domain. The other palindrome containing protein is cellular human tumor suppressor p53. It contains palindrome in its beta-sheet domain that interacts with palindromic fragment of DNA. One example of such protein is systemin, an 18-amino-acid-long peptide. A great number (26%) of different protein palindromes were found. One can ask the questions, Do palindromes occur in protein, and if so, what function they play? We have searched the protein SWISSPROT database for palindromic sequences. 
Palindromes in telomeres are crucial for initiation of replication. 
For example, restriction enzymes often recognize palindromic sequences of DNA. They occur in genomes of all organisms and have various functions. Palindromes in DNA consist of nucleotides sequences that read the same from the 5'-end to the 3'-end, and its double helix is related by twofold axis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
